Kirsten Clinical Research
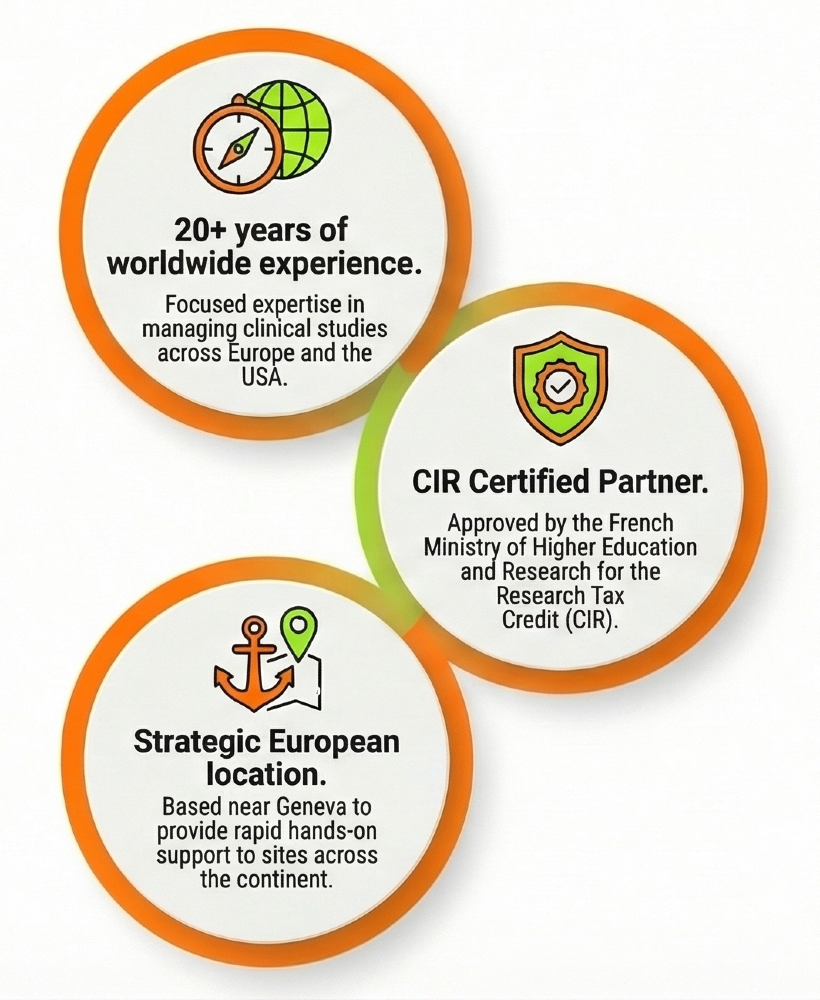
Kirsten Clinical Research is a trusted partner for the coordination and management of clinical trials. Accredited by the French Ministry of Higher Education and Research under the Research Tax Credit (CIR), the company has been supporting sponsors and study partners since 2011.
Based near Geneva and backed by more than 20 years of operational experience, your studies are conducted with rigor, close collaboration, and strong commitment across a wide range of therapeutic areas, with a strong expertise in rare diseases.
We provide independent, tailor-made consultancy built on trust, clarity, and seamless collaboration. Kirsten Imbert works hand in hand with sponsors and study partners, adapting the support to each project’s needs while ensuring strict respect for timelines, budgets, and current quality standards. My approach is rooted in proximity, adaptability, and operational excellence.
Your projects will be a success thanks to our:
- Strong commitment to advancing medical research
- Energy and momentum, driven by hands‑on involvement and rigorous work
- Solutions‑oriented approach, turning complexity into clear, actionable steps
- Strong ability to connect people and bring teams into alignment
- Clear and responsive communication
- Solid professionalism, ensuring quality and integrity, including in “rescue” situations
Expertise
Founded and led by Kirsten Imbert – van Harmelen, who brings more than 20 years of experience in research and clinical study operations across the world, the company offers expertise in clinical trial coordination, project management, monitoring, quality assurance, and recruitment and retention strategies.
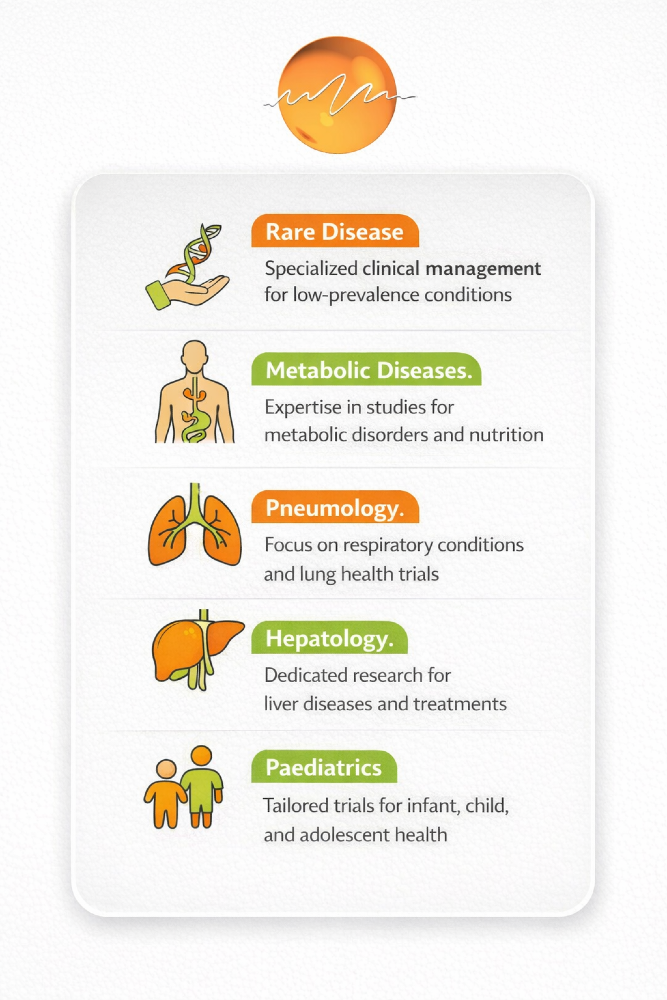
Operational and Therapeutic Expertise
- Phase I-IV, drug, nutrition research, and medical device trials.
- Metabolic diseases,
- Endocrinology,
- Hepatology,
- Pneumology,
- Inflammatory diseases,
- Orthopedics, Neurosurgery,
- Haematology
- Cardiovascular.
Scientific Background - Kirsten Imbert
I graduated from Wageningen University in the Netherlands with a Master’s degree in Nutrition and Health Sciences, specializing in both Nutrigenomics and Toxicology. This strong scientific foundation supports my rigorous, analytical approach to clinical research. Over the past 20 years, I have contributed to the successful development of multiple drugs, medical devices and medical food products, supporting studies conducted across Europe and the United States, and have been instrumental in the development of several products that are now on the market.
Approach
I am often called upon to step into complex or stalled projects, re-establish collaboration among stakeholders, clarify priorities, and restore progress, particularly in participant recruitment and site engagement, with clarity and momentum. My approach is people-centered and solution-oriented. I connect stakeholders, build trust quickly, and turn complex situations into clear priorities and actionable next steps. When timelines stall or communication breaks down, I step in to restore collaboration and move studies forward with structure, transparency, and efficiency.